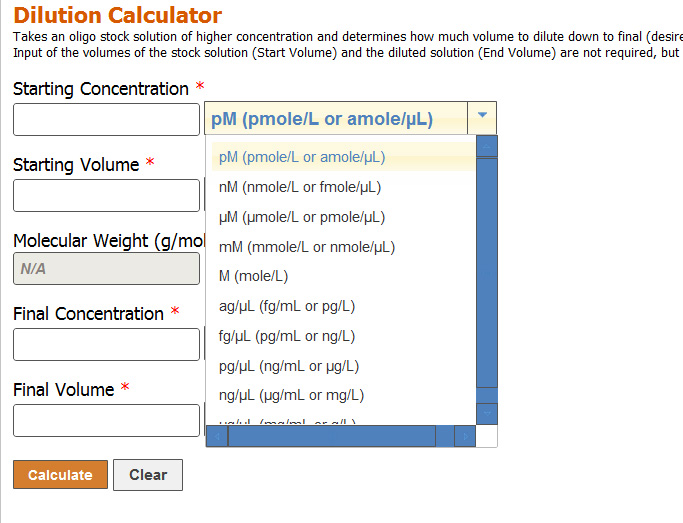
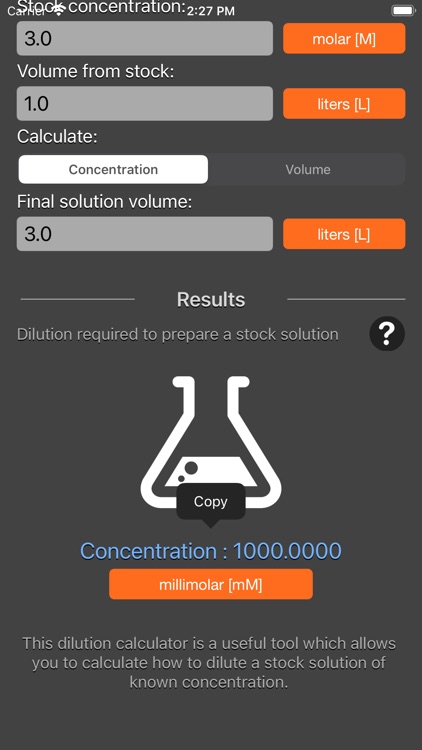
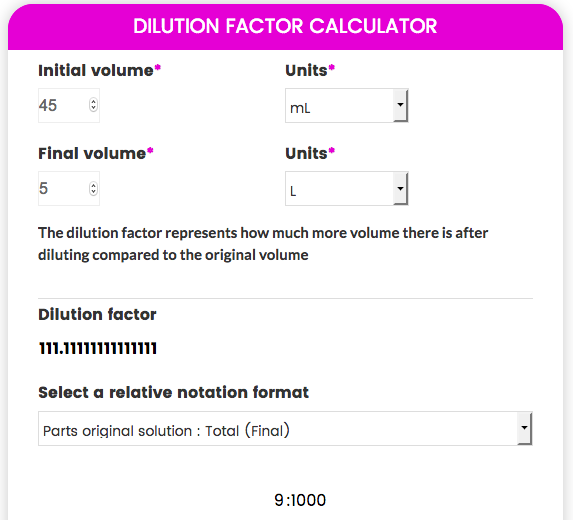
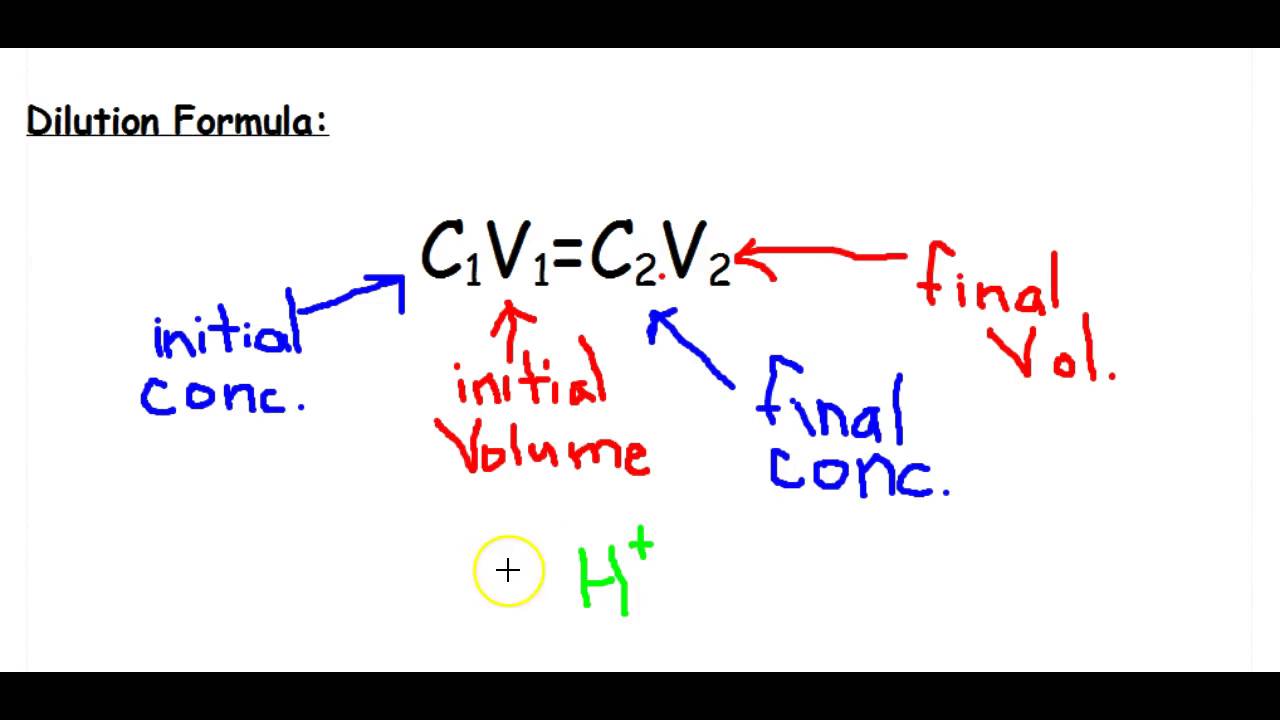
Solutions:- Part 1 - Solutions Preparation used in Clinical Laboratory, and Dilution Formula - Labpedia.net

Dilutions. Solve problems involving the dilution of solutions. Include: dilution of stock solutions, mixing common solutions with different volumes and. - ppt download

Other units of concentration. Dilution Equation MV = MV Molarity (volume) before dilution = molarity (volume) after dilution How many liters of 12 M H. - ppt download