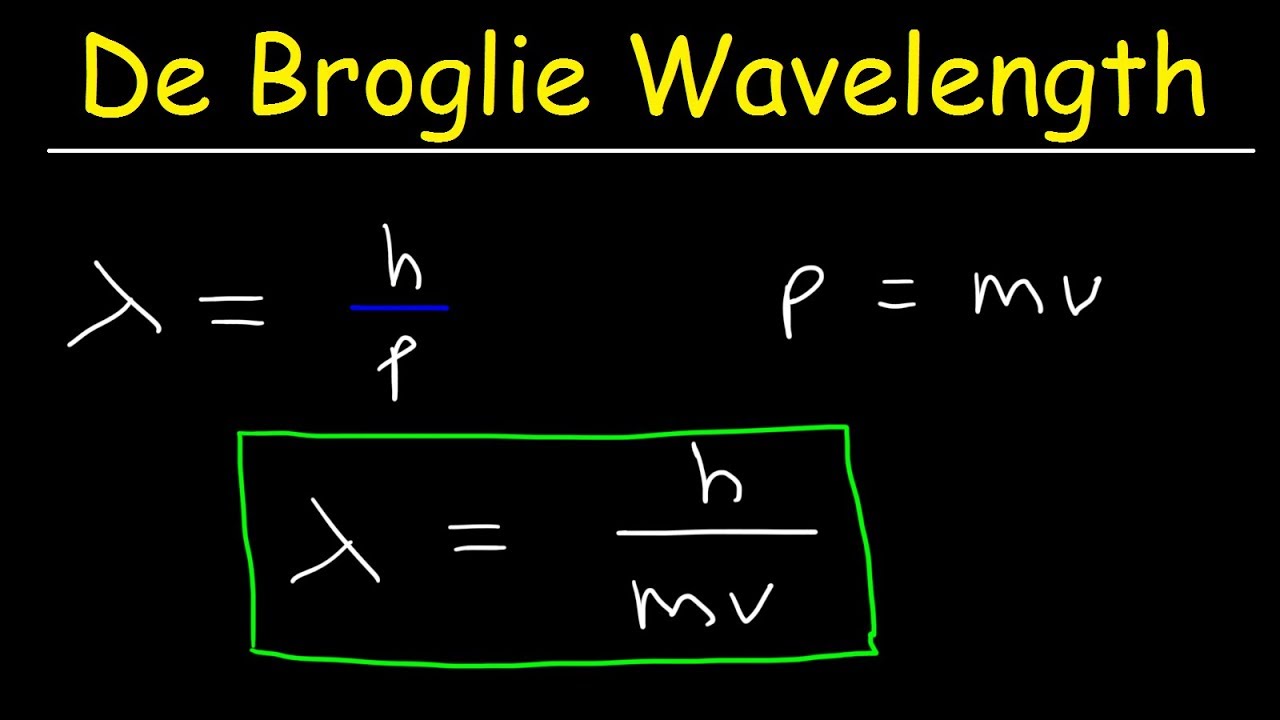
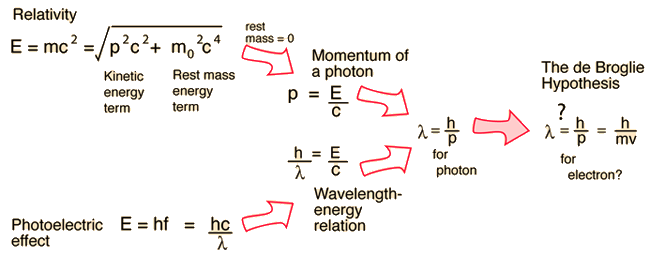
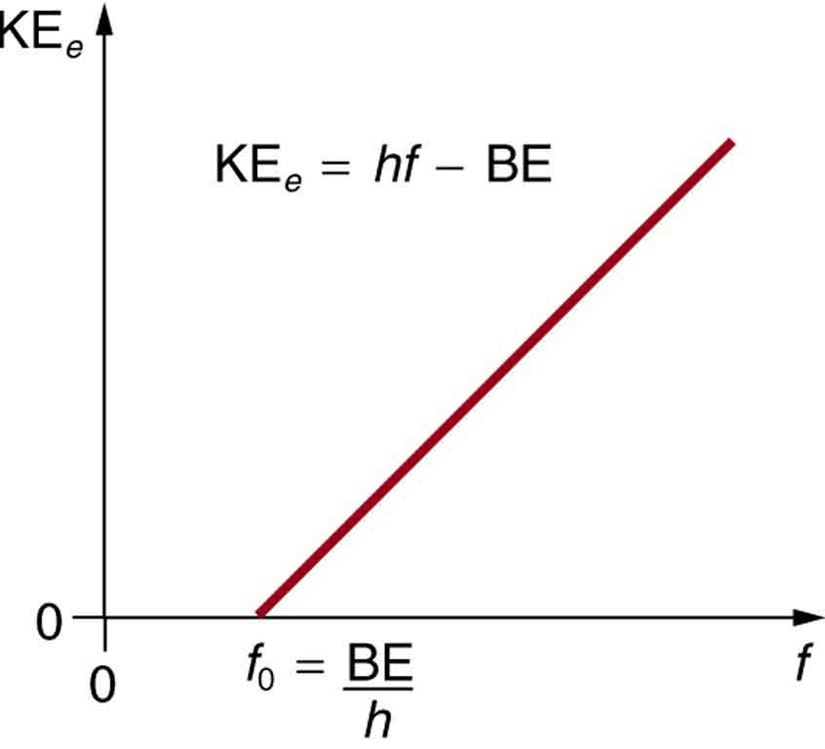
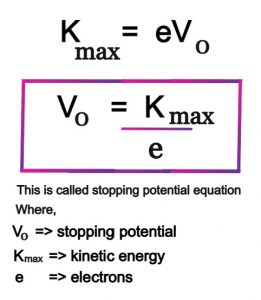
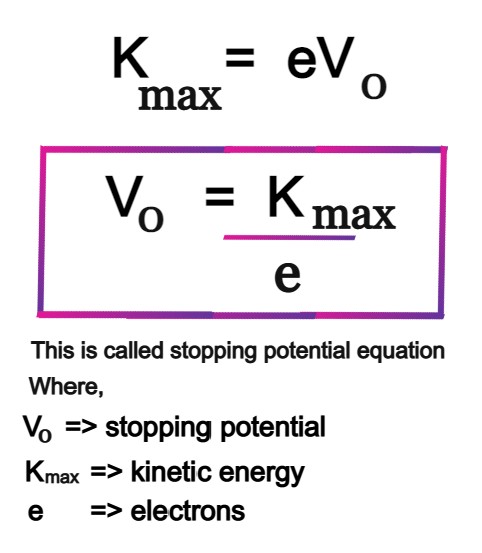
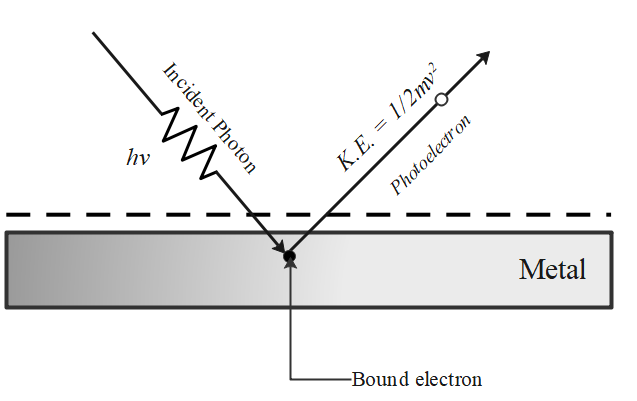
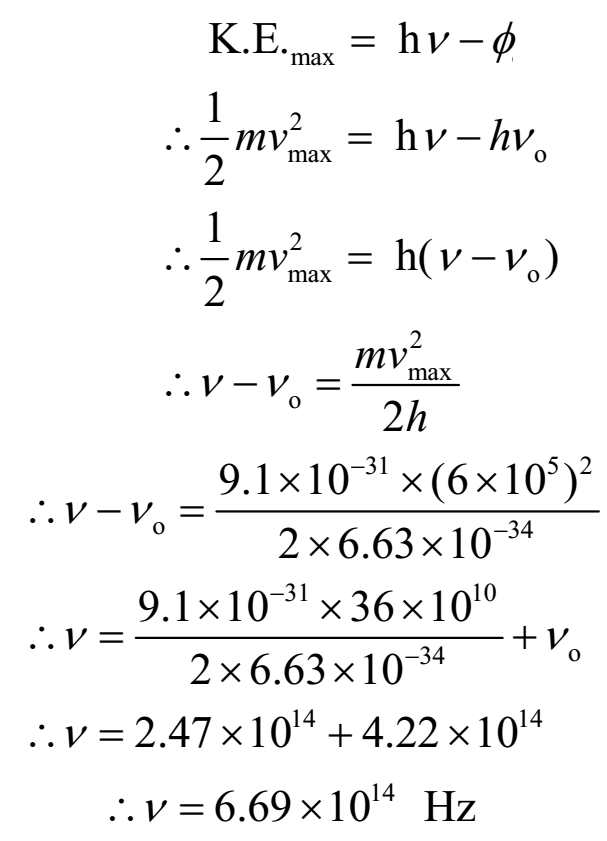The ejection of the photoelectron from the silver metal in the photoelectric effect experiment can be stopped by applying the voltage of 0.35 V when the radiation 256.7 nm is used. Calculate
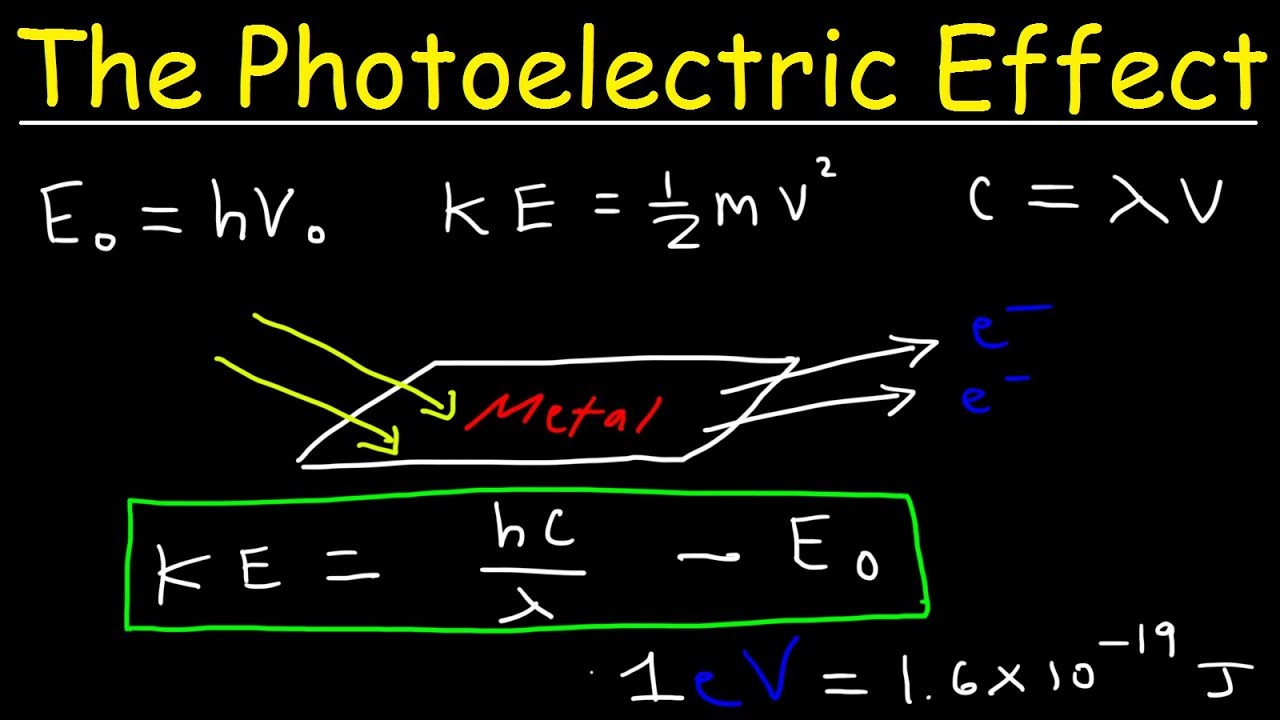
Photoelectric Effect, Work Function, Threshold Frequency, Wavelength, Speed & Kinetic Energy, Electr - YouTube

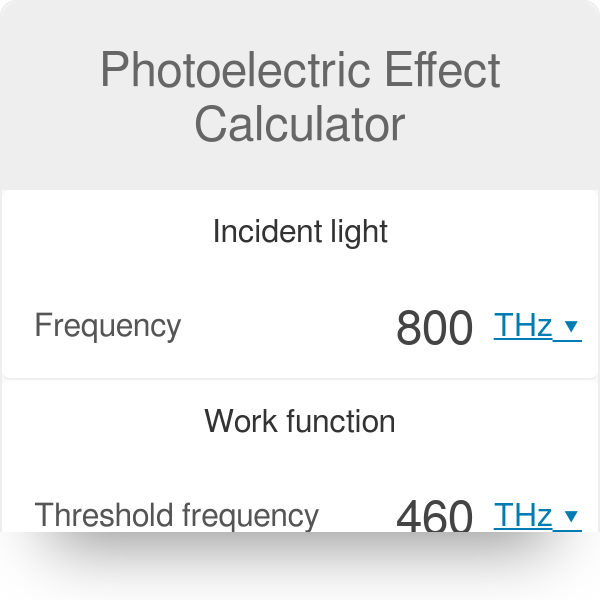
Calculate the threshold frequency of photon for photoelectric emission from a metal of work - YouTube

In a photoelectric effect experiment, irradiation of a metal with light of frequency 5.2 × 10^14sec^-1 yields electrons with maximum kinetic energy 1.3 × 10^-19 J . Calculate the v0 of the metal.
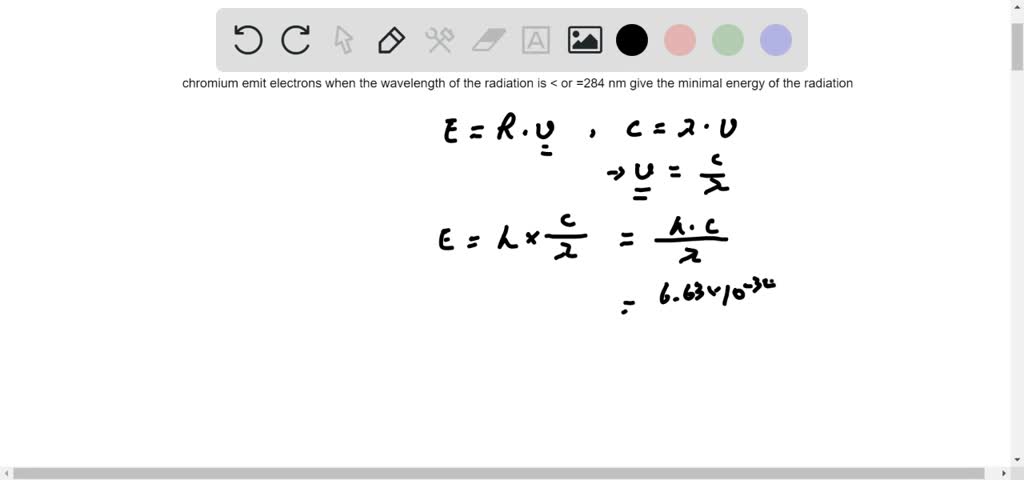
SOLVED:Applying Models In discussions of the photoelectric effect, the minimum energy needed to remove an electron from the metal is called the threshold energy and is a characteristic of the metal. For

Finding the Average Velocity of Electrons Emitted in a Photoelectric Experiment with Given Incident Light Frequency and Material Work Function | Physics | Study.com