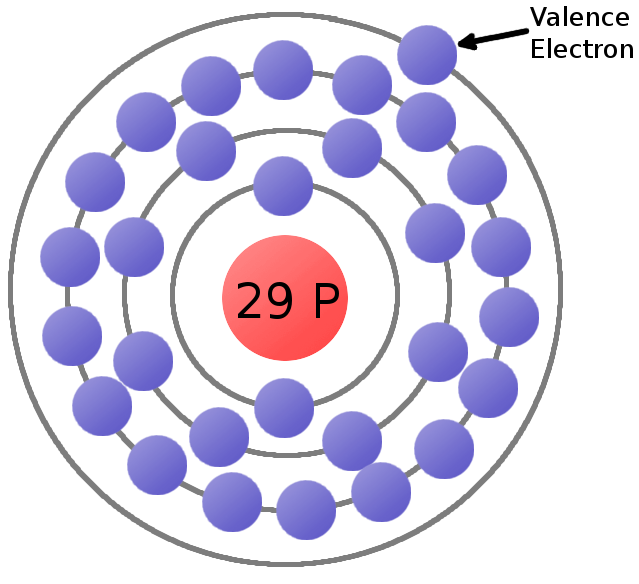
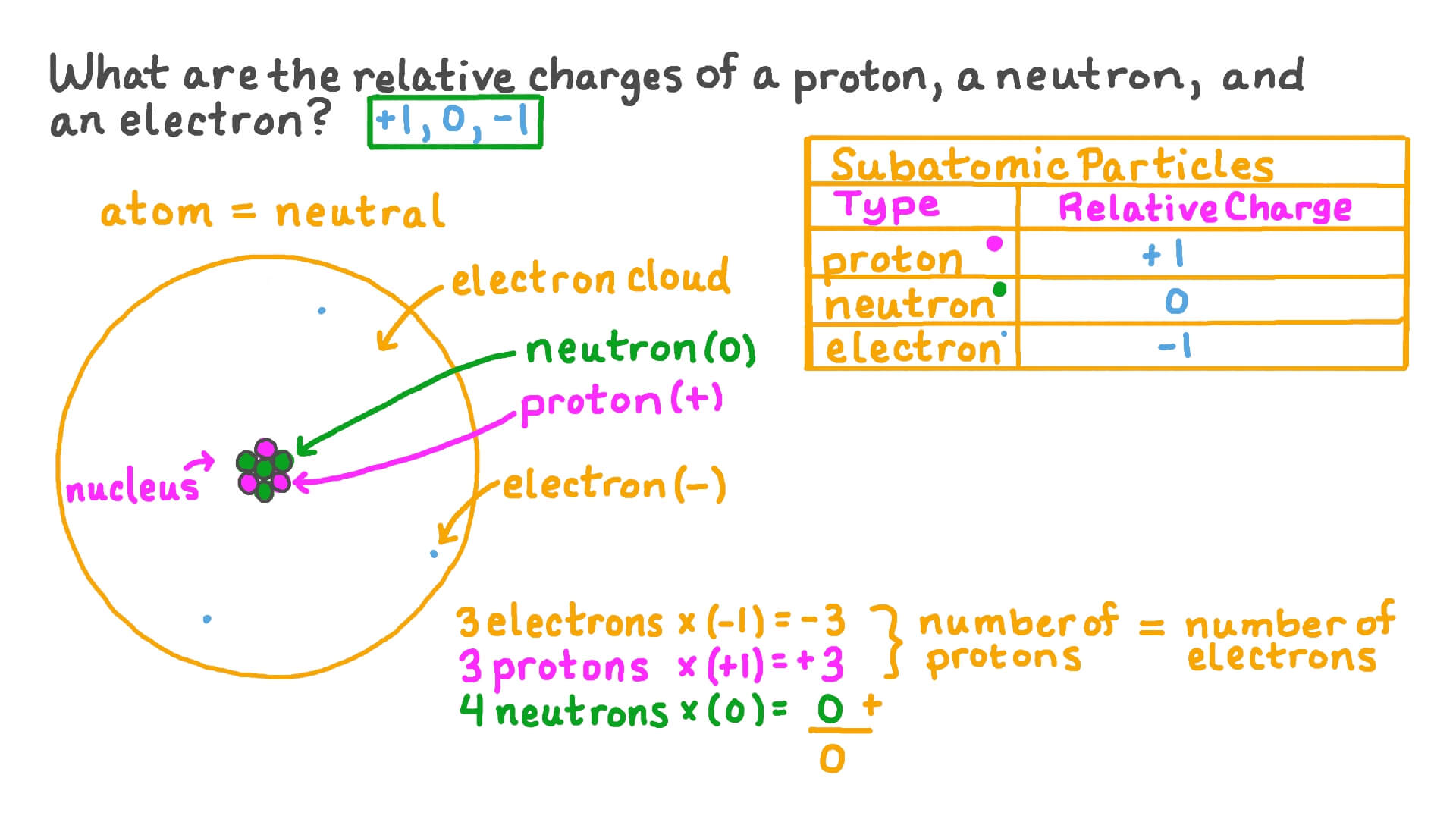
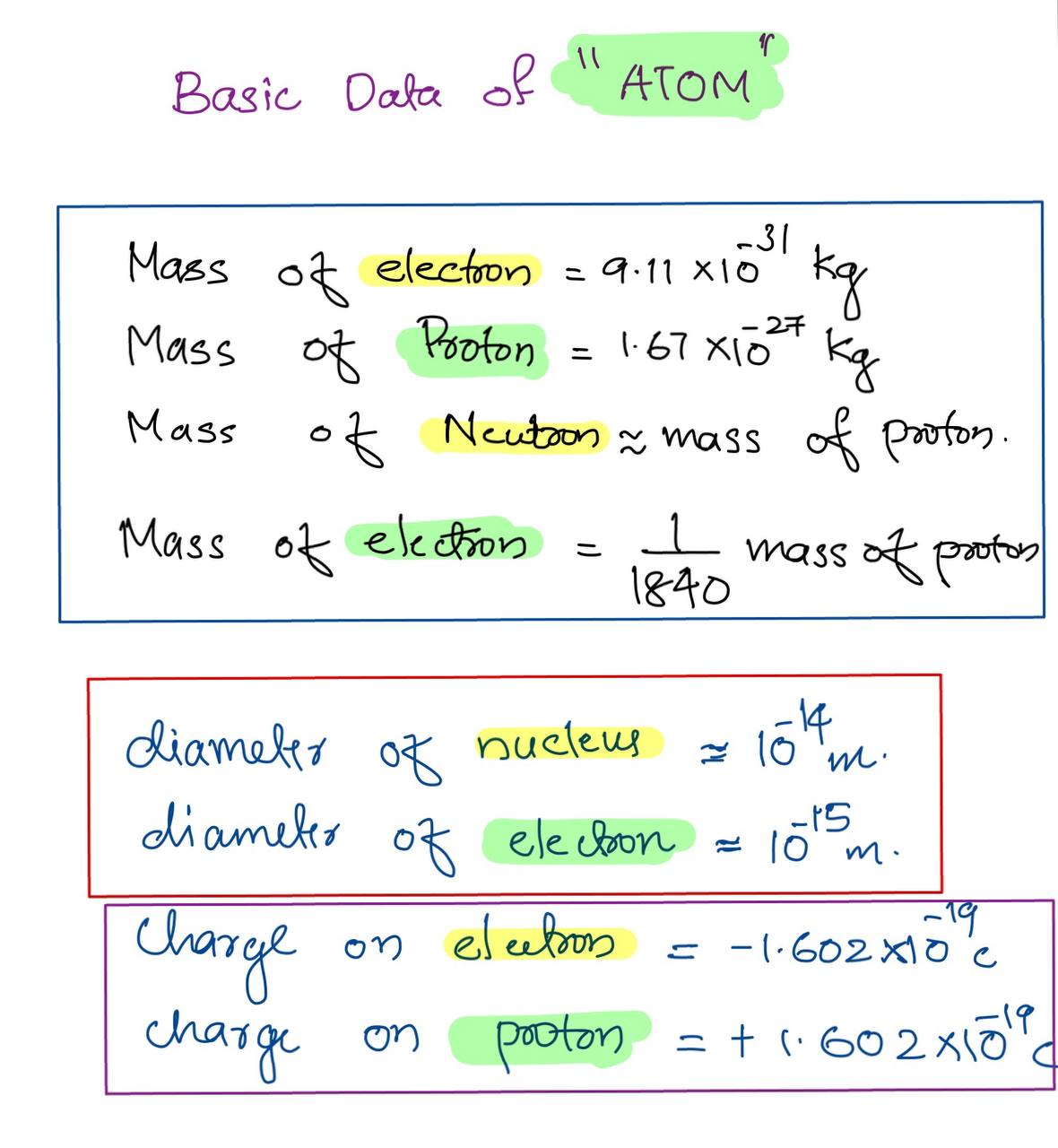
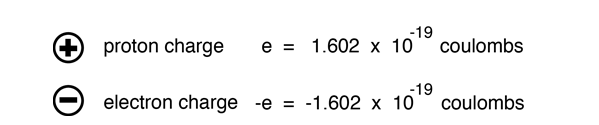How to Calculate Total Charge in Coulombs of an Arrangement of Protons and Electrons | Physics | Study.com

How to Determine the Number of Electrons on an Object Given its Net Charge in Coulombs | Physics | Study.com

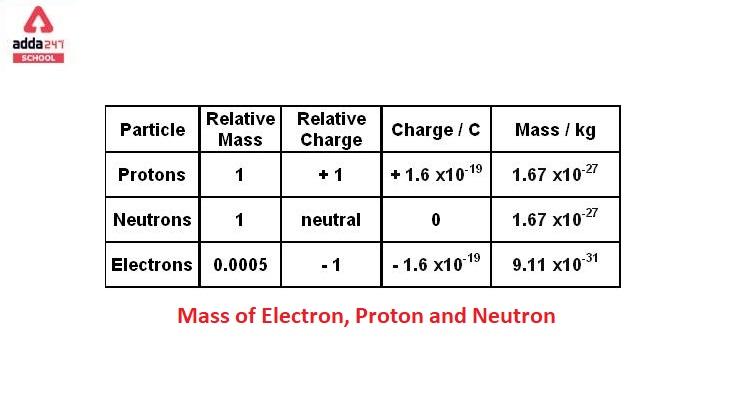
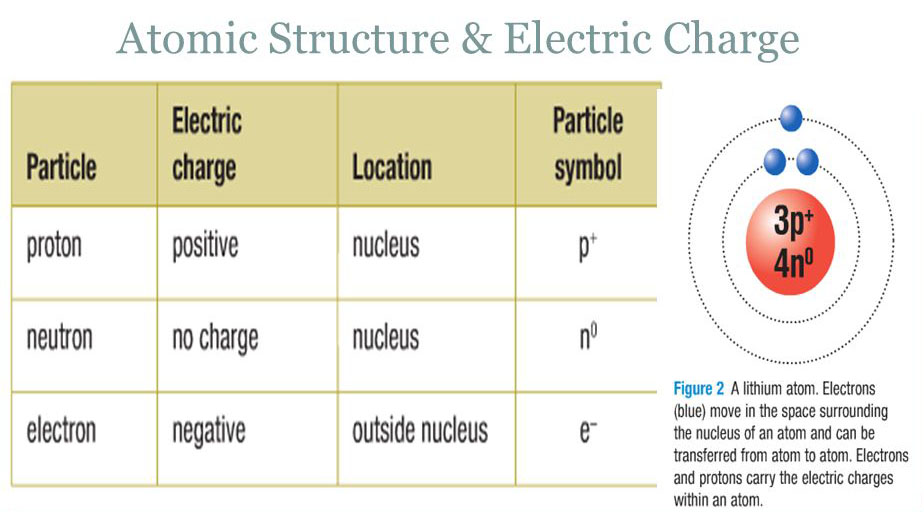
The charge on a proton is +1.6 X 10(-19) C and that on an . electron is -1.6 X 10 (-19)C. Does it mean that the electron . has a charge 3.2

The charge of an electron is `1.6 xx 10^(-19)C` what will be the value of charge on `Na^(+)` ion. - YouTube